Association and linkage analysis of COL1A1 and AHSG gene polymorphisms with femoral neck bone geometric parameters in both Caucasian and Chinese nuclear families1
Introduction
Osteoporosis is a systemic skeletal complex disease characterized by bone loss and decreased bone strength, leading to increased fracture risk. It is a major health problem both in Caucasians and in Chinese. Currently, over 200 million people worldwide suffer from this disease[1].
Bone strength as the ultimate measurement of resistance to osteoporotic factures (OF) is determined by bone mineral density (BMD), bone geometry, bone microarchitecture, and the quality of bone material[2–4]. Bone geometry can significantly improve the prediction of osteoporosis or OF risk[5–7]. Most research have found that women with femoral neck (FN) fractures have longer hip axis length, larger FN-shaft angle than controls without fractures[8,9]. However, Gnudi et al found no specificity of bone geometric parameters for spine fracture risk[8]. In addition, bone geometric parameters as complex traits were under strong genetic determination[10,11].
It is well known that hip fracture is the most serious and disabling type of OFs[12]. The ethnic difference of hip fracture risk is well known[13,14]. Almost all of the studies consistently reported that the age and sex-adjusted annual rate of hip fracture was higher in Caucasians than in Asians[13,14]. The ethnic differentiation of bone geometry may be partially responsible for the above observations. For example, smaller bone widths and a shorter hip axis length were detected in women of African origin compared with Caucasians, and these observed racial differences could contribute to an approximately 25% decrease in the risk of hip fracture among blacks[15]. However, a few studies have focused on the different bone geometry between Asians and Caucasians, and whether these ethnic differences of bone geometry come from the ethnic-specific genetic determination remains unknown.
Genes encoding the alpha 1 chain of collagen type 1 (COL1A1) and alpha 2-HS-glycoprotein (AHSG) are 2 important genes, which might influence bone geometry. COL1A1 is one of most abundant proteins in bone matrix. Mutations in COL1A1 and collagen type I alpha2 (COL1A2) genes were estimated to be responsible for up to 90% of cases of the Mendelian disease named osteogenesis imperfecta, which is associated with a very low bone mass and an increased fracture risk. A G/T polymorphism affecting the Sp1 binding site in the first intron of the COL1A1 gene, also known as rs1800012 or g1546 G>T, was regarded as a predictor of reduced BMD and increased the risk of fragility fractures, reported in several studies[16,17]. However, the importance of the COL1A1 gene on bone phenotypes may not be explained by the Sp1 polymorphism in Chinese, as the Sp1 polymorphism was absent in Chinese population[18]. Another important mutation in the promoter region of COL1A1 gene, the PCOL2 variance (rs1107946 or g-1997 G>T), was first found by Garcia-Giralt et al in postmenopausal women of Spanish origin[19]. Garcia-Giralt et al established a significantly association between PCOL2 variance and BMD variations at the lumbar spine and FN[19]. Meanwhile, a strong interaction between PCOL2 and Sp1 variance was observed in a study of elderly Caucasian females[20]. In light of foregoing studies, the PCOL2 polymorphism was selected as a valuable genetic marker to explain the importance of COL1A1 gene in Chinese.
AHSG as a human plasma glycoprotein has a high level of serum and mineralized bone[21]. AHSG can bind Ca2+ with high affinity to prevent apatite formation in the circulation[22], and may play an important role in bone cell metabolism by influencing the recruitment of osteoclastic precursors to bone[23] and modulating bone resorption[24]. The AHSG phenotypes, characterized by ACG (Thr)/ATG (Met) at amino acid position 230 at exon 6 and ACC (Thr)/AGC (Ser) at amino acid position 238 in exon 7, have been proven to contribute to the genetic influence of lumbar vertebral BMD, femoral neck BMD[25], and calcaneal broadband ultrasound attenuation[26]. The SacI polymorphism (rs4918 or g238 C>G) represents the nucleotide substitution of C–G at amino acid position 238 in exon 7, which may change the charge of molecules and generate the relevant mobility on isoelectric focusing.
To date, only a few studies have focused on the comparison of bone geometric parameters across different races[27,28], but no genetic study has simultaneously investigated the contribution of bone candidate genes on the variation of bone geometric parameters in both Caucasians and Chinese. Thus, in the present study, we first compared bone geometric parameters of FN between the Caucasian and Chinese population and then tested the effect of the AHSG and COL1A1 genes on the variation of bone geometric parameters of FN in both Caucasians and Chinese, our study would contribute to understand the various genetic determination of bone geometry between different ethnic populations.
Materials and methods
Subjects The study was supported by Creighton University (Omaha, USA) and Hu-nan Normal University (Changsha, China). The Caucasian sample consisted of 605 Caucasian individuals from 157 nuclear families aged from 19 to 87 years. All Caucasian individuals came from the city of Omaha in the USA. The family size ranged from 3 to 12 with a mean of 4.2, yielding 372 sibling pairs. The Chinese sample was composed of 400 nuclear families with a total of 1228 individuals aged from 19 to 80 years. Each nuclear family was composed of both parents and at least 1 healthy premenopausal offspring with the average family size of 3.14, yielding 57 sibling pairs. All the subjects belonged to the Chinese Han group and were recruited from the city of Shanghai in China. Before participating in the 2 projects, each subject signed an informed consent document. Both populations were collected using the same excluded criterion to minimize the potential confounding factors on osteoporosis studies[29].
Phenotyping BMD and bone area of the FN were measured by dual energy X-ray absorptiometry (DXA). Both machines were calibrated daily. The short-term precision in vivo for femoral BMD and bone size was 1.87% and 1.94%, as well as 0.80% and 0.06% in Caucasians and Chinese, respectively. In the Hologic systems, the width of the FN region of interest (ROI) was standardized at 1.5 cm, and W was the average FN periosteal diameter obtained by dividing the FN area by 1.5 cm. We used the FN BMD and W to estimate 5 indices of the FN geometry: (1) cross sectional area (CSA) –– the area with mineralized bone tissue, excluding bone marrow space; (2) cortical thickness (CT); (3) endocortical diameter (ED) –– an estimate of medullary bone thickness; (4) section modulus (Z) –– a measure of bending strength, which is consistent with the skeleton load and implicates in hip fragility; and (5) buckling ratio (BR) –– an index of bone geometric instability, which indicates the risk of fracture by buckling[30,31].
Genotyping Genomic DNA was isolated from the peripheral blood in Caucasians using a commercial isolation kit (Gentra Systems, Minneapolis, MN, USA) following the procedure detailed in the kit. In the Chinese, the total genomic DNA was isolated using the phenol-chloroform extraction method. The genotyping procedure is the polymerase chain reaction and restriction fragment length polymorphism (PCR-RFLP). The PCR reaction mixed was as follows: 0.1–0.3 µg genomic DNA, 0.2–0.24 mmol/L dNTP (Promega Biotech, Madison, WI, USA), 1 unit of Taq polymerase (Promega Biotech, Madison, WI, USA or Sangon Co, Shanghai, China), respective proportions of MgCl2, oligonucleotide primers and 10×enzyme buffer in a total volume of 25 µL. The PCR was carried out on either a PE9700 Thermal Cycler (Perkin Elmer Cetus, Norwalk, CT, USA) or an Omnigene Thermocycler (Hybaid, Ashford, UK). After the amplification, 8 µL aliquots of products were digested with the respective restriction endonucleases for 3–12 h at 37 °C and subjected to electrophoresis on 2% or 3% agarose gel in 1×TAE buffer (Tris-Acetate-EDTA) stained with ethidium bromide and detected under UV light. The information of genotyping the 3 polymorphisms are presented in Table 1.
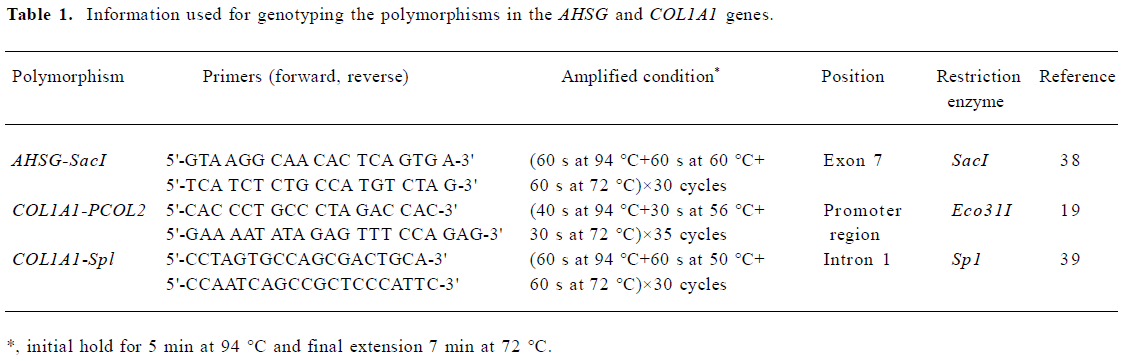
Full table
Statistical analyses We used the quantitative transmission disequilibrium test program (QTDT)[32] to test population stratification, total family association, within-family association, and linkage between the AHSG-SacI, COL1A1-PCOL2 and Sp1 polymorphisms, and 5 bone geometric parameters. An orthogonal model in the QTDT analysis decomposed the genotype score into orthogonal between family (βb) and within family (βw) components. The βb was specific to each nuclear family and could be sensitive to the population structure, but the βw was significant only in the presence of both linkage disequilibrium (LD) and linkage. The within-family association only evaluated the βw, but the total family association evaluated the association at the population level on all the subjects. Therefore, the total family association may produce false positive results in the presence of population stratification/admixture. The QTDT can also test population stratification by evaluating if the βb is equal to βw. The above tests were based on a variance component framework using the maximum likelihood ratio computed with the QTDT. One thousand permutation tests using a Monte-Carlo permutation framework were used to generate the empirical P value to assess the reliability of within-family association. SPSS (version11, Chicago, IL, USA) t-tests were used to compare the difference of unadjusted and adjusted (by age, height, weight, and gender) bone geometric parameters in both randomly selected offspring and parent subgroups. All of the phenotypic data were tested for normality by the Kolmogorov-Smirnov test implemented in the software Minitab (Minitab Inc, State College, PA, USA) before the association and linkage tests. No significant deviation from the normal distribution was found for the phenotypic data analyzed.
Results
The basic characteristics of the 2 population groups were summarized in Table 2. Generally, the Chinese have significant lower CSA, CT, ED, and Z, compared with Caucasians. The t-test in the offspring showed the significant differences of all bone geometric phenotypes (except ED) between Caucasians and Chinese using both unadjusted and adjusted (by age, height, weight, and gender) data, while in the parent group, all 5 geometric phenotypes detected significant differences after being adjusted by age, height, weight, and gender (P<0.01). The allele frequencies of three polymorphisms were presented in Table 3. The allele frequency of the AHSG-SacI polymorphism in the Chinese was lower than in the Caucasians.
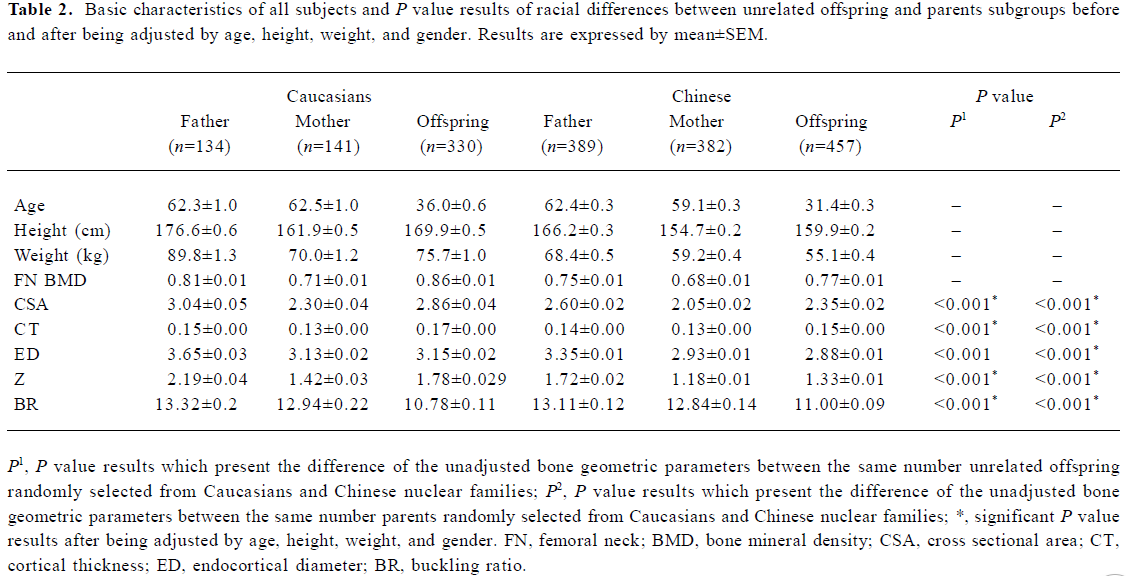
Full table
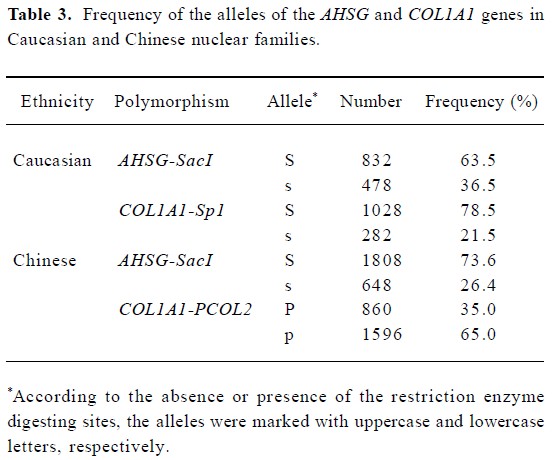
Full table
The results of the population stratification, within-family association, and linkage test were shown in Table 4. The significant population stratification in Caucasians may have influenced the results of total family association (data not shown). In Caucasians, we found significant within-family association for COL1A1-Sp1 polymorphism with CSA, CT, ED, BR (P=0.018, 0.002, 0.023, and 0.001, respectively), and significant linkages were detected with BR (P=0.039). In Chinese population, the within-family associations between the COL1A1-PCOL2 polymorphism and CT and BR were significant (P=0.012 and 0.008, respectively). Although the AHSG-SacI polymorphism within-family association was observed with CSA and CT in Caucasians, evidence of linkage were detected for CT and BR (P=0.042 and 0.014, respec-tively). However, in Chinese population, neither association nor linkage results were found for the AHSG-SacI polymorphism. After one thousand permutation tests, P<0.009 and P<0.008 were required for an individual test to achieve a global significance level of 0.05 in Caucasians and Chinese, respectively. The within-family association for COL1A1-Spl polymorphism with CSA, CT, ED, and BR were still significant in Caucasians (P=0.005, <0.001, 0.006, and <0.001, respectively). In the Chinese, the association for COL1A1-PCOL2 polymorphism with BR, CSA, CT, and ED was significant or nominally significant (P=0.002, 0.08, 0.014, and 0.05, respectively) after permutation.
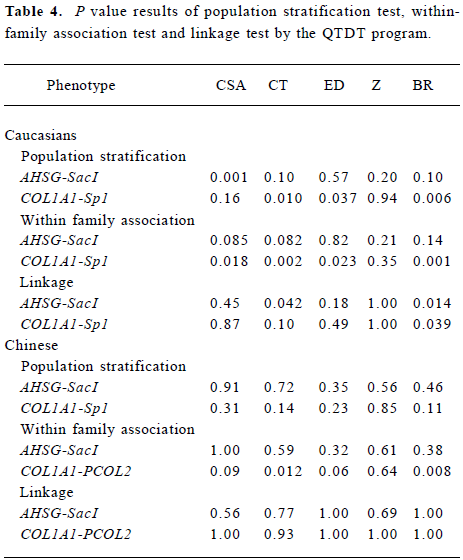
Full table
Discussion
Our major findings in this study are as follows: (1) there is a differential bone geometry between Chinese and Caucasians; and (2) the COL1A1 gene may be associated with the variations of the FN bone geometric parameters in both population groups, and the AHSG gene may be linked to the variations of the FN bone geometric parameters in Caucasians, but not in Chinese. These results may provide some suggestive clues on the ethnic-differential genetic determination of bone geometry in Caucasians and Chinese, that is, the 2 ethnic populations may both share some common and different genes regulating the variation of bone geometry. This study represents our first efforts in investigating the importance of the COL1A1 and AHSG genes on bone geometry in both Caucasians and Chinese.
Our differential bone geometry between the 2 ethnic populations was partly consistent with another study in Caucasian and Chinese subjects aged 18–93 years[33], but we failed to detect evidence of significant racial difference on ED in young female groups. These conflicting results may be the difference in sampling methods, covariates used for the adjustment, or some other confounding issue. A review summarized the ethnic difference in osteoporosis-related phenotypes between Caucasians and Asians and its potential underlying genetic determination. The potential genetic evidence included the different heritability and inheritance mode of bone phenotypes, the different osteoporosis candidate genes, and the differential results in related molecular studies between them[13,14].
The significant within-family association between the polymorphisms of the COL1A1 gene and bone geometry suggests that the polymorphisms are likely to be in linkage disequilibrium with a nearby functional mutation, or the polymorphisms themselves may have important effects on the variation of bone geometry in both Caucasians and Chinese. Simultaneously, the consistent within-family association across ethnic populations implicates that Caucasians and Chinese may share the same effects of the COL1A1 gene on regulating the variation of bone geometry.
The AHSG gene is linked to FN bone geometric parameters in Caucasians, but not in Chinese. The results were similar as previous studies, in which within Chinese population no evidence was found regarding the association between the AHSG gene and BMD[34]. Two factors may explain the inconsistent linkage. First, the inconsistent linkage may be due to the differential linkage power. The power of linkage analysis is higher in Caucasians than in Chinese. In the Caucasian sample, the family size ranged from 3 to 12 with a mean of 4.2, yielding 372 sib pairs, whereas in the Chinese sample, despite the large number of nuclear families (400), only 57 sib pairs were informative for the linkage analyses (since the majority of families had only 1 daughter), rendering only modest power to detect the true linkage. Second, the ethnic-specific genetic determination for bone geometry may be responsible for the inconsistent linkage. A possible explanation is that there is a functional mutation influencing bone geometry nearby the AHSG gene on the same chromosome in Caucasians, but not in Chinese. Interestingly, Xiong et al recently found linkage evidence for 3q27 with CSA and CT where the AHSG gene is located in Caucasians[35].
Our results may support previous findings that the COL1A1 gene may be a predictor of osteoporosis and OF risk in Caucasians[16,36]. We found significant within-family associations between 4 FN geometric parameters (CSA, CT, ED, and BR) and the COL1A1-Sp1 polymorphism in Caucasians. Our further analysis found that the ss genotype groups had higher BR than those with other genotypes. Therefore, from the present results, we suppose that the different genotypes of the COL1A1 gene in subjects regulate the diversity of bone geometry, which will eventually result in differential OF risk. Further genetic and molecular researches are needed to confirm this hypothesis.
There were some potential limitations in our study. All 5 FN bone geometric parameters were calculated based on the DXA-derived BMD and ROI. DXA which only provided a 2-dimensional analysis was used to estimate the approximation of the 3-dimensional structure of bone, thus the calculated bone geometric parameters may not be suitable for reflecting 3-dimensional bone structure features. The mean values of BMD from the conventional Hologic ROI for FN (1.5 cm wide) are on average 14% lower than Beck’s mean values of BMD from the narrow neck region[37]. This difference may result in the lower estimates of CSA, CT, and higher estimates of ED and BR in our study.
In summary, our results suggest that the COL1A1 gene may play a role in the FN bone geometric variation in both Caucasians and Chinese, while the influence of the AHSG gene may be different in the 2 ethnic groups. The understanding of ethnic-specific genetic determination for bone geometry will assist in understanding the pathogenesis of osteoporosis comprehensively and develop ethnic-specific approaches for the prevention, diagnosis, and treatment of osteoporosis.
References
- Reginster JY, Burlet N. Osteoporosis: a still increasing prevalence. Bone 2006;38:S4-9.
- Ammann P, Rizzoli R. Bone strength and its determinants. Osteoporos Int 2003;14 Suppl 3:S13-8.
- Bouxsein ML. Bone quality: where do we go from here? Osteoporos Int 2003;14 Suppl 5:118-27.
- Pulkkinen P, Partanen J, Jalovaara P, Jamsa T. Combination of bone mineral density and upper femur geometry improves the prediction of hip fracture. Osteoporos Int 2004;15:274-80.
- Looker AC, Beck TJ. Maternal history of osteoporosis and femur geometry. Calcif Tissue Int 2004;75:277-85.
- Peacock M, Turner CH, Econs MJ, Foroud T. Genetics of osteoporosis. Endocr Rev 2002;23:303-26.
- Gnudi S, Ripamonti C, Gualtieri G, Malavolta N. Geometry of proximal femur in the prediction of hip fracture in osteoporotic women. Br J Radiol 1999;72:729-33.
- Gnudi S, Malavolta N, Testi D, Viceconti M. Differences in proximal femur geometry distinguish vertebral from femoral neck fractures in osteoporotic women. Br J Radiol 2004;77:219-23.
- Malavolta N, Frigato M, Mule R, Ripamonti C. Femoral neck morphology differentiates femoral neck from vertebral or hip osteoporotic fracture. Reumatismo 2003;55:93-7. Italian..
- Koller DL, Liu G, Econs MJ, Hui SL, Morin PA, Joslyn G, et al. Genome screen for quantitative trait loci underlying normal variation in femoral structure. J Bone Miner Res 2001;16:985-91.
- Slemenda CW, Turner CH, Peacock M, Christian JC, Sorbel J, Hui SL, et al. The genetics of proximal femur geometry, distribution of bone mass and bone mineral density. Osteoporos Int 1996;6:178-82.
- Kannus P, Parkkari J, Sievanen H, Heinonen A, Vuori I, Jarvinen M. Epidemiology of hip fractures. Bone 1996;18:57S-63S.
- Ross PD, Norimatsu H, Davis JW, Yano K, Wasnich RD, Fujiwara S, et al. A comparison of hip fracture incidence among native Japanese, Japanese Americans, and American Caucasians. Am J Epidemiol 1991;133:801-9.
- Lei SF, Chen Y, Xiong DH, Li LM, Deng HW, Yan L, et al. Ethnic difference in osteoporosis-related phenotypes and its potential underlying genetic determination. J Musculoskelet Neuronal Interact 2006;6:36-46.
- Theobald TM, Cauley JA, Gluer CC, Bunker CH, Ukoli FA, Genant HK. Black-white differences in hip geometry. Study of Osteoporotic Fractures Research Group. Osteoporos Int 1998;8:61-7.
- Mann V, Ralston SH. Meta-analysis of COL1A1 Sp1 polymorphism in relation to bone mineral density and osteoporotic fracture. Bone 2003;32:711-7.
- Ralston SH, Uitterlinden AG, Brandi ML, Balcells S, Langdahl BL, Lips P, et al. Large-scale evidence for the effect of the COLIA1 Sp1 polymorphism on osteoporosis outcomes: the GENOMOS study. PLoS Med 2006;3:e90.
- Lei SF, Deng FY, Liu XH, Huang QR, Qin Y, Zhou Q, et al. Polymorphisms of four bone mineral density candidate genes in Chinese populations and comparison with other populations of different ethnicity. J Bone Miner Metab 2003;21:34-42.
- Garcia-Giralt N, Nogues X, Enjuanes A, Puig J, Mellibovsky L, Bay-Jensen A, et al. Two new single-nucleotide polymorphisms in the COL1A1 upstream regulatory region and their relationship to bone mineral density. J Bone Miner Res 2002;17:384-93.
- Liu PY, Lu Y, Long JR, Xu FH, Shen H, Recker RR, et al. Common variants at the PCOL2 and Sp1 binding sites of the COL1A1 gene and their interactive effect influence bone mineral density in Caucasians. Med Genet 2004;41:752-7.
- Dickson IR, Poole AR, Veis A. Localisation of plasma alpha2HS glycoprotein in mineralising human bone. Nature 1975;256:430-2.
- Jahnen-Dechent W, Schinke T, Trindl A, Muller-Esterl W, Sablitzky F, Kaiser S, et al. Cloning and targeted deletion of the mouse fetuin gene. J Biol Chem 1997;272:31496-503.
- Malone JD, Teitelbaum SL, Griffin GL, Senior RM, Kahn AJ. Recruitment of osteoclast precursors by purified bone matrix constituents. J Cell Biol 1982;92:227-30.
- Yang F, Schwartz Z, Swain LD, Lee CC, Bowman BH, Boyan BD. Alpha 2-HS-glycoprotein: expression in chondrocytes and augmentation of alkaline phosphatase and phospholipase A2 activity. Bone 1991;12:7-15.
- Dickson IR, Gwilliam R, Arora M, Murphy S, Khaw KT, Phillips C, et al. Lumbar vertebral and femoral neck bone mineral density are higher in postmenopausal women with the alpha 2HS-glycoprotein 2 phenotype. Bone Miner 1994;24:181-8.
- Zmuda JM, Eichner JE, Ferrell RE, Bauer DC, Kuller LH, Cauley JA, et al. Genetic variation in alpha 2HS-glycoprotein is related to calcaneal broadband ultrasound attenuation in older women. Calcif Tissue Int 1998;63:5-8.
- Nelson DA, Pettifor JM, Barondess DA, Cody DD, Uusi-Rasi K, Beck TJ. Comparison of cross-sectional geometry of the proximal femur in white and black women from Detroit and Johannesburg. J Bone Miner Res 2004;19:560-5.
- Yan L, Crabtree NJ, Reeve J, Zhou B, Dequeker J, Nijs J, et al. Does hip strength analysis explain the lower incidence of hip fracture in the People’s Republic of China? Bone 2004;34:584-8.
- Deng HW, Shen H, Xu FH, Deng HY, Conway T, Zhang HT, et al. Tests of linkage and/or association of genes for vitamin D receptor, osteocalcin, and parathyroid hormone with bone mineral density. J Bone Miner Res 2002;17:678-86.
- Duan Y, Beck TJ, Wang XF, Seeman E. Structural and biomechanical basis of sexual dimorphism in femoral neck fragility has its origins in growth and aging. J Bone Miner Res 2003;18:1766-74.
- Beck TJ, Stone KL, Oreskovic TL, Hochberg MC, Nevitt MC, Genant HK, et al. Effects of current and discontinued estrogen replacement therapy on hip structural geometry: the study of osteoporotic fractures. J Bone Miner Res 2001;16:2103-10.
- QTDT program [updataed by 2006 Jun, cited by 2006 Jul]. Available from: http://www.sph.umich.edu/csg/abecasis/QTDT/
- Wang XF, Duan Y, Beck TJ, Seeman E. Varying contributions of growth and ageing to racial and sex differences in femoral neck structure and strength in old age. Bone 2005;36:978-86.
- Liu XH, Liu YJ, Jiang DK, Li YM, Li MX, Qin YJ, et al. No evidence for linkage and/or association of human alpha2-HS glycoprotein gene with bone mineral density variation in Chinese nuclear families. Calcif Tissue Int 2003;73:244-50.
- Xiong DH, Shen H, Xiao P, Guo YF, Long JR, Zhao LJ, et al. Genome-wide scan identified QTLs underlying femoral neck cross-sectional geometry that are novel studied risk factors of osteoporosis. J Bone Miner Res 2006;21:424-37.
- Mann V, Hobson EE, Li B, Stewart TL, Grant SF, Robins SP, et al. A COL1A1 Sp1 binding site polymorphism predisposes to osteoporotic fracture by affecting bone density and quality. J Clin Invest 2001;107:899-907.
- Beck TJ, Looker AC, Ruff CB, Sievanen H, Wahner HW. Structural trends in the aging femoral neck and proximal shaft: analysis of the Third National Health and Nutrition Examination Survey dual-energy X-ray absorptiometry data. J Bone Miner Res 2000;15:2297-304.
- Osawa M, Umetsu K, Ohki T, Nagasawa T, Suzuki T, Takeichi S. Molecular evidence for human alpha 2-HS glycoprotein (AHSG) polymorphism. Hum Genet 1997;99:18-21.
- Grant SF, Reid DM, Blake G, Herd R, Fogelman I, Ralston SH. Reduced bone density and osteoporosis associated with a polymorphic Sp1 binding site in the collagen type I alpha 1 gene. Nat Genet 1996;14:203-5.
