Effect of GNTI, a kappa opioid receptor antagonist, on MK-801-induced hyperlocomotion and stereotypy in mice1
Introduction
Neuropeptides and their receptors play an important role in modulating the functioning of the brain[1]. Opioids and opioid receptors take part in multiple physiological processes both within the central nervous system and in the periphery. Opioid receptors are divided into 3 major types: kappa, mu and delta receptors[2]. Mu and delta opioid receptors are considered the major binding sites for endorphins and enkephalins, while kappa opioid receptors are considered to bind to dynorphins with the highest affinity.
The kappa opioid system has been shown to interact with other neurotransmitter systems, including the glutamate system and the dopamine system. At the neuroanatomic level, kappa opioid receptors have been shown to localize on the dopaminergic and glutamatergic nerve terminals in the dorsal and ventral striatum, and their activation inhibits dopamine (DA) and glutamate release[3]. In particular, using microdialysis to measure the dopamine levels in the nucleus accumbens, the basal extracellular dopamine levels were decreased by stimulating the kappa opioid receptor in wild-type mice, and the deletion of the dynorphin gene in the knockout mice resulted in reduced levels of dopamine[4], suggesting the involvement of the kappa opioid system on dopamine neurotransmission. Also, using the rodent model of Parkinson’s disease with selective dopamine neuron depletion by 6-hydroxydopamine treatment, pretreatment with a kappa opioid receptor selective agonist potentiated the dopamine neuron loss[5], again suggesting the kappa opioid participation in dopamine transmission. Furthermore, by direct electrophysiological recording in midbrain dopamine neurons, it was found[6] that the activation of the kappa opioid receptor directly inhibited some dopamine neurons in the brain, and this kappa receptor-mediated inhibition was blocked by the kappa receptor antagonist nor-BNI, which was structurally similar to GNTI [5'-guanidinyl-17-(cyclo-propylmethyl)-6,7-dehydro-4,5α-epoxy-3,14-dihydroxy-6,7-2',3'-indolomorphinan][7].
Interaction between the kappa opioid system and the glutamate system is also known. Over-stimulation of NMDA (N-methyl-D-aspartate) receptors can lead to seizure, and activation of the kappa opioid receptor provided an anticonvulsive effect in the 3 rat models of experimental seizures[8,9], suggesting a neuroprotective effect of the kappa opioid system via its effect on the glutamate neurotransmission path-way. On the other hand, the kappa opioid agonist-produced anticonvulsant effect could be antagonized by a glycine/NMDA receptor antagonist[10], suggesting a reciprocal interaction between the kappa opioid system and the gluta-mate system.
Because of the interactions between the kappa opioid system and the dopamine and glutamate systems, it has been suggested that the kappa opioid receptor could serve as a potential drug target for the development of therapeutic agents for schizophrenia and drug abuse[11]. In support of this view, Royston and colleagues used [3H]U-69593 autoradiograms and reported that the kappa receptor distribution in schizophrenic patients displayed a pattern different from patients without the condition[12]. However, they lack studies that directly examine the effect of modulating kappa opioid receptors in experimental models relevant for psychosis and schizophrenia. The present study was designed to address this issue by using a selective kappa opioid antagonist in a well-established rodent model for psychotic symptoms of schizophrenia.
Opioid receptors interact with a variety of ligands, including endogenous peptides, opiates, and numerous synthetic compounds with different structural scaffolds[13,14]. For kappa opioid receptors however, highly selective ligands are rather limited. GNTI is a selective kappa opioid receptor antagonist, as it displays several hundred-fold selectivity over mu and delta opioid receptors[15–17]. This was the selective kappa antagonist of choice for the present study.
MK-801 (dizocilpine maleate) is a non-competitive NMDA receptor antagonist, and is known to cause increased locomotion and various stereotypical behavior in rodents[18]. The induction of the NMDA receptor hypofunction by MK-801 is considered a pharmacological model for psychotic symptoms of schizophrenia, as it mimics various aspects of psychopathology observed in this illness[19–21]. To explore the potential role of the kappa opioid receptor in modulating brain functions involved in psychosis and schizophrenia, we chose this rodent model and report the findings.
Materials and methods
Chemicals (+)-MK-801 and GNTI were from Tocris Cookson, Inc (Ellisville, MO, USA). MK-801 was dissolved in saline (0.9% NaCl solution) as master stock solution (1 mg/mL), and aliquots were kept frozen at -70 oC until use. The master stock solution of GNTI (4 mg/mL) was similarly prepared and stored. The doses of MK-801 used in our experiments were 0.3 mg/kg and 0.6 mg/kg. The doses of GNTI were 0.1 mg/kg, 0.3 mg/kg, 1.0 mg/kg, 3.0 mg/kg and 10.0 mg/kg. Before each experiment, aliquots of master stocks were thawed and diluted with saline to the final concentrations for systemic administration by ip injection. Dilutions were designed to give a final injection volume of 100 µL per animal for each dose of MK-801 or GNTI administered.
Animals Male BALB/c mice aged about 5 weeks, weighing 20–22 g were obtained from the Shanghai Laboratory Animal Center, Chinese Academy of Sciences (Warrant N
Apparatus The locomotor activity of mice was measured with a photobeam activity system for open field configuration (San Diego Instruments Inc, San Diego, CA, USA) consisting of 10 chambers. Each chamber contained a cage (45 cm in length, 25 cm in width and 20 cm in height). Horizontal motion was detected by 8 photobeams lengthwise spaced at 5 cm apart, and 4 photobeams widthwise spaced at 4.5 cm apart, with the entire photobeam array positioned at a height of 2 cm above ground level. Locomotion measurements were obtained by automated detection of beam crossings. Centered at the ceiling over every activity chamber was a mounted video camera to record behavior on videotapes for offline analysis.
Behavior measurements Since studying a full dose range required several days, experiments were conducted over sequential days, with daily experimental session conducted between 8:00–15:00.
The animals were placed individually in a chamber, and locomotion measurements were obtained by automated detection of beam crossings. For stereotypy measurements, the animals in the locomotion measurement chambers were videotaped for the 4 h recording period. Hence, for each animal, stereotypy and locomotion data represented concurrent behavior, but were independent of each other. Stereotypy measurements were presented as the percentage of time spent in stereotypical behavior. Stereotypy was broadly defined and included a number of abnormal movements including head weaving, rotation, rolling and ataxic behavior. We chose to include ataxia into the broadly-defined stereotypy measurements rather than scoring ataxia separately, because our earlier studies have clearly demonstrated that by using a single parameter of broadly defined stereotypy, the outcome was readily interpretable and was not at all affected differentially by varying amount of ataxia[19]. The stereotypy measurement was scored for the first 2 min out of each 10 min time segment. The following experiments were conducted:
(1) 60 mice were divided into 6 groups, with each group containing 10 mice. The animals were ip injected with the appropriate dose of saline or GNTI, then placed immediately into the recording chamber for behavioral measurements; (2) 70 mice were divided into 7 groups, with each group containing 10 mice. The mice received 2 ip injections; the first injection was with saline or the appropriate dose of GNTI, and then after 30 min, the second injection was saline or 0.3 mg/kg MK-801. The mice were then placed immediately into the recording chamber for behavioral measurements; and (3) was the same as (2), except that the dose of MK-801 was 0.6 mg/kg.
Data analysis Data were analyzed using the software GraphPad Prism 4.0. All data were subjected to statistical analysis. Averaged data are shown with error bars indicating the SEM. The effects of the different doses of GNTI at the same dose of MK-801 were compared with controls using one-way analysis of variance (ANOVA) with Dunnett’s post-hoc test. Significance is ascribed for P<0.05.
Results
GNTI inhibition of MK-801-induced hyperlocomotion MK-801 is known to dose-dependently modulate locomotion in mice[19,20]. To characterize any effect that GNTI may exert on MK-801-induced locomotor activities, we first examined the effect of GNTI administered alone in mice. As shown in Figure 1A, systemic injection of GNTI alone showed a mild inhibitory effect, especially during the initial 120 min, suggesting that GNTI itself may exert a mild impact on locomotion. It should be noted, however, that this effect appeared to be rather limited as the cumulative locomotion data for the entire recording period (Figure 1B) showed that there was no statistical difference for any of the GNTI dose compared with the saline control (F5,52=4.690, P=0.0013).

We next examined GNTI effect on MK-801-induced hyperlocomotion. We chose 2 doses of MK-801 to study: 0.3 mg/kg (Figure 1C) and 0.6 mg/kg (Figure 1D), because our previous study suggested that these 2 doses of MK-801 appeared to represent a transition point for the bimodal effects of MK-801 on locomotion and stereotypy[19]. In that study, while at 0.3 mg/kg MK-801 primarily increased locomotion, with low-level stereotypy (well below 50%, even at the peak time), 0.6 mg/kg MK-801 was able to induce substantial stereotypy; the peak stereotypy reached about 90% of the maximum level (Figure 2A[19]), thus indicating that 0.3 mg/kg vs 0.6 mg/kg MK-801 may bridge a transition point. In our current study, GNTI appeared to display an inhibitory effect on MK-801-induced hyperlocomotion (Figures 1C,1D).
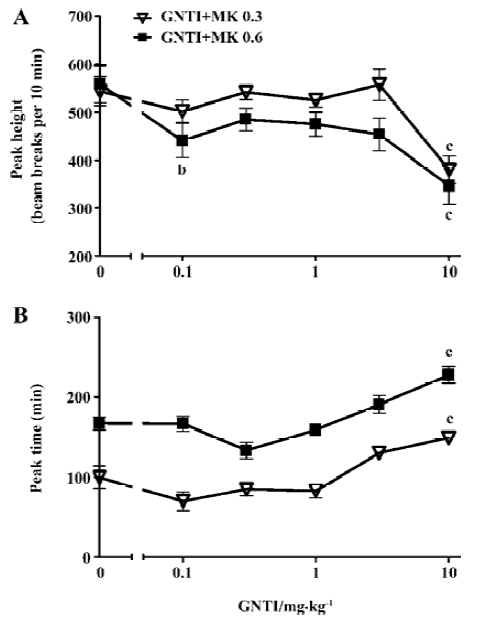
MK-801-induced hyperlocomotion peaks were further analyzed to examine GNTI effect on these parameters. As shown in Figure 2A, GNTI reduced peak height, reaching statistical significance at higher doses (GNTI+MK 0.3 groups: F6,62=25.64, P<0.0001; GNTI+MK 0.6 groups: F6,62=13.12, P<0.0001). Time to reach the hyperlocomotion peak was delayed by GNTI, again reaching statistical significance at higher GNTI doses (GNTI+MK 0.3 groups: F6,62=16.79, P<0.0001; GNTI+MK 0.6 groups: F6,59=50.05, P<0.0001). These data indicate that GNTI displayed a mild inhibitory effect on MK-801-induced hyperlocomotion, with statistically significant inhibition only at higher GNTI doses (Figure 2B).
GNTI effects on MK-801-induced stereotypy Aside from induction of hyperlocomotion, MK-801 also results in stereotypy, and the ability of various drugs to modulate MK-801-induced locomotion and stereotypy has become a preclinical test for screening new antipsychotics. We thus also examined GNTI effect on MK-801-induced stereotypy. We manually scored stereotypy by watching videotapes taken during locomotion measurements, and the data are summarized in Figure 3. The control groups (GNTI alone or saline alone) did not show any stereotypy, while MK-801 resulted in an appreciable amount of stereotypical behavior. Figure 3A shows the percentage of time spent in stereotypy by mice receiving 0.3 mg/kg MK-801, and GNTI dose-dependently inhibited MK-801-induced stereotypy. In particular, MK-801-induced stereotypy was almost completely inhibited by 10 mg/kg GNTI. In fact, of the 10 mice in the 10 mg/kg GNTI group, only 3 out of 10 mice displayed very slight stereotypy, and the other 7 mice showed no stereotypy behavior at all. The total amount of time spent in stereotypy in the 10 mg/kg GNTI group was not significantly different from the saline control group, again indicating a complete inhibition of stereotypy by 0.3 mg/kg MK-801. To better quantify the GNTI effect on MK-801-induced stereotypy, the cumulative stereotypy was calculated and shown in Figure 3B. These cumulative data confirmed that GNTI was able to dose-dependently inhibit stereotypy induced by 0.3 mg/kg MK-801, showing significant effect (F5,53=94.95, P<0.0001) between the GNTI groups and the MK-801 control group (saline+0.3 mg/kg MK-801).

When GNTI effect was examined for mice receiving 0.6 mg/kg MK-801, a rather different profile was observed. As shown in Figure 3C, only the highest GNTI dose (10 mg/kg) appeared to show appreciable inhibition of stereotypy. Indeed, this was confirmed by the quantitative analysis of cumulative stereotypy (Figure 3D). Only the 10 mg/kg GNTI group showed statistical difference from that of the MK-801 control (saline+0.6 mg/kg MK-801; F5,48=9.032, P<0.0001), while other GNTI doses did not show significant difference from the MK-801 controls.
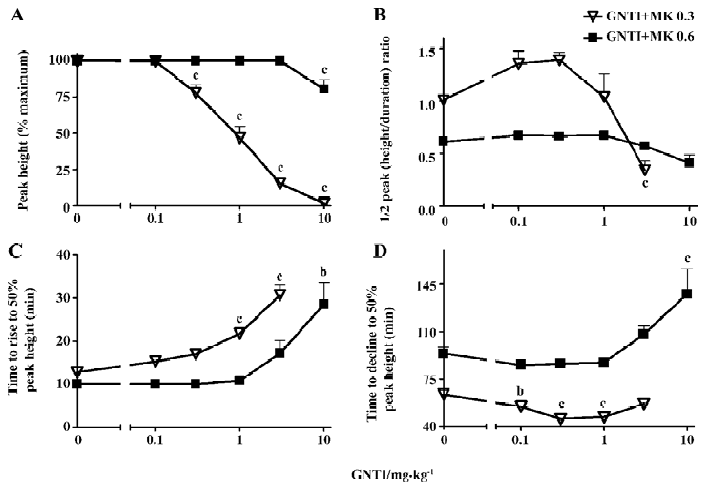
To better explore and highlight the dynamic properties that GNTI exerted on MK-801-induced stereotypy, we analyzed various parameters of the stereotypy peak. First, we examined the stereotypy peak height and calculated as a percentage of the MK-801 alone control, which was the maximum peak height ever reached. As shown in Figure 4A, GNTI was able to dose-dependently reduce the percentage peak height of stereotypy by 0.3 mg/kg MK-801 across most GNTI doses, and was only effective at the highest dose when MK-801 was 0.6 mg/kg (GNTI+MK 0.3 groups: F5,53=96.21, P<0.0001; GNTI+MK 0.6 groups: F5,48=8.604, P<0.0001). To examine the overall peak shape, we used a composite parameter of 1/2 peak (height/duration) ratio, which was calculated by dividing the 1/2 peak height with the 1/2 peak duration (the time duration from the rising phase 50% point to the declining phase 50% point). In essence, this composite parameter reflected the relative peak shape by giving a numerical ratio between the peak’s height over its width. As shown in Figure 4B, this parameter of peak shape differentiated between 0.3 mg/kg vs 0.6 mg/kg MK-801 for the impact of GNTI on stereotypy. While the 1/2 peak (height/duration) ratio remained unchanged relative to the no GNTI control for 0.6 mg/kg MK-801, it appeared to display a bell-shaped dose-response curve for 0.3 mg/kg MK-801. Next, we further examined the dynamic properties of the stereotypy peak by taking the 50% peak height as a reference point, and separately analyzed the rising phase and the declining phase of the stereotypy peak for their respective time to reach this reference point of 50% peak height. For the rising phase of the stereotypy peak, the time for stereotypy to rise to 50% peak height was delayed by GNTI (Figure 4C), with a more pronounced effect for 0.3 mg/kg MK-801 and some effect for 0.6 mg/kg MK-801 (GNTI+MK 0.3 groups: F4,44=23.28, P<0.0001; GNTI+MK 0.6 groups: F5,48=9.704, P<0.0001). For the declining phase of the peak, we looked at the time for stereotypy to decline to 50% peak height (Figure 4D), and GNTI at several doses (0.1 mg/kg, 0.3 mg/kg and 1.0 mg/kg) actually resulted in a faster decline compared to the 0.3 mg/kg MK-801 control (F4,43=9.166, P<0.0001), while for the 0.6 mg/kg MK-801 groups, GNTI at the highest dose of 10 mg/kg resulted in a slower decline (F5,47=6.458, P<0.0001). Thus, for the 0.6 mg/kg MK-801 groups, both the time to rise to 50% peak height and the time to decline to 50% peak height were delayed by 10 mg/kg GNTI (Figures 4C,4D). On the other hand, for the 0.3 mg/kg MK-801 groups, GNTI delayed the rising phase kinetics (Figure 4C) while hastening the declining phase kinetics (Figure 4D). These results again highlighted the differential effects that GNTI exerted on 0.3 mg/kg vs 0.6 mg/kg MK-801-induced stereotypy.
Discussion
MK-801 is a non-competitive NMDA receptor antagonist and is known to cause increased locomotion and various stereotypical behavior in rodents. These MK-801-induced behaviors are commonly used as a model for preclinical evaluation of psychoactive properties of drugs including schizophrenia. GNTI is a selective antagonist for the kappa opioid receptor[16,17,22]. There is evidence for interaction between the kappa opioid receptor and MK-801-mediated NMDA receptor[23], and there have been reports of potential involvement of the kappa opioid receptor in schizophrenia[12,24]. Considering these reports, we undertook the current study and examined the effect that GNTI exerted on MK-801-induced behavior.
Our laboratory has previously studied the effect of MK-801 on locomotion and stereotypy and reported the MK-801 effect over a rather wide range of concentrations (from 0.02 mg/kg to 10 mg/kg) in mice[19,20]. Based on our previous results, we chose 2 doses of MK-801 for the current study: 0.3 and 0.6 mg/kg MK-801. This is because that these 2 doses of MK-801 appeared to represent a transition point for the bimodal effects of MK-801 on locomotion and stereotypy[19], while 0.3 mg/kg MK-801 primarily increased locomotion with much reduced stereotypy (well below 50% even at the peak time) and 0.6 mg/kg MK-801 was able to induce substantial stereotypy (peak time about 90%).
Our data indicate that GNTI displayed an inhibitory effect on MK-801-induced hyperlocomotion (Figures 1C, 1D), by reducing the hyperlocomotion peak height and delaying the time to reach the hyperlocomotion peak (Figure 2). Interestingly, the influence of GNTI on MK-801-induced stereotypy showed a differential effect between 0.3 mg/kg vs 0.6 mg/kg MK-801 (Figure 3). For 0.3 mg/kg MK-801, GNTI inhibited stereotypy in a dose-dependent manner (Figures 3A,3B). For 0.6 mg/kg MK-801, however, GNTI was largely ineffective except at the highest dose of 10 mg/kg (Figures 3C,3D). This differential effect was also evident when examining the hyperlocomotion peak height; the peak height by 0.3 mg/kg MK-801 was progressively reduced more and more as the GNTI dose became higher, whereas the peak height by 0.6 mg/kg MK-801 was only significantly affected by the highest dose of GNTI (Figure 4A). The overall peak shape parameter, 1/2 peak (height/duration) ratio (Figure 4B) also showed the divergent trend for the 2 doses of MK-801. While this parameter for 0.3 mg/kg MK-801 showed a bell-shaped GNTI dose-response curve, for 0.6 mg/kg MK-801 it stayed relatively unchanged from the control group value (Figure 4B). These results suggest that 0.3 mg/kg and 0.6 mg/kg MK-801 may indeed represent 2 different phases of MK-801-induced behavior, and such differential sensitivity was readily observed when the effect of GNTI on stereotypy was examined. In addition, the pharmacodynamic aspect of GNTI may represent a contributing factor in the differential dosage effect, as GNTI is structurally related to nor-BNI[7] and nor-BNI has been shown to have rather long acting dura-tion[25]. It should be noted that, our broad definition of stereotypy included ataxia-like behavior. In our previous study, it was shown that including vs not including ataxic behavior in stereotypy scoring would not affect the outcome of MK-801-induced stereotypy[19]. Nonetheless, there is a possibility that ataxia may constitute a confounding factor in animals’ response to drug effect, and should be taken into consideration when viewing the outcome of this study.
Generally, hyperlocomotion and stereotypy in animal experiments are considered to be 2 behavioral signs reflective of schizophrenia symptoms. Using longer recording periods and observing stereotypy in addition to locomotion as a behavioral measure, appears to be particularly important for studying modulation of MK-801 effects by other drugs[19]. The mechanisms by which MK-801 increases locomotion and stereotypy in a spontaneous alternation paradigm appear to involve glutamatergic-dopaminergic, glutamatergic-serotonergic or dopaminergic-serotonergic interactions[26]. Regarding the role of neuropeptide and opioid involvement in schizophrenia, there are 2 schools of thoughts: one maintains that endogenous opioid peptides are in deficit in schizophrenia, thus supplementing them may be helpful in treating schizophrenia, and the other believes that the neuropeptides are already too high in schizophrenic patients[27]. The results of our study indicate that antagonizing kappa opioid receptors can reverse MK-801-induced effect on behavior. These results, if extrapolated to human conditions, would suggest that reducing the activity of kappa opioid receptors may be beneficial for psychotic symptoms in schizophrenics. Other groups have reported that the deletion of the kappa-opioid receptor increased dialysate DA levels in the nucleus accumbens, suggesting the existence of a tonically active kappa opioid receptor-mediated system that inhibits the basal activity of mesoaccumbal DA neurons[4].
Nor-BNI is another selective antagonist for kappa opioid receptors[28], and is structurally related to GNTI[7]. Nor-BNI pretreatment blocks stress-induced potentiation of cocaine-related conditioned place preference[29], but nor-BNI fails to reverse MK-801 on the development of morphine tolerance[30] and the PPI disruption mediated by both apomorphine and MK-801[31]. Nor-BNI could weaken behavioral withdrawal caused by electrical stimulation of the nucleus paragigantocellularis[32]. For the first time, our study utilized GNTI as a selective antagonist to probe kappa opioid receptor involvement in the mouse model of MK-801-induced behavior, and provided conceptual support for considering GNTI or similar kappa opioid antagonists as antipsychotic drugs. Our study also demonstrated the usefulness of GNTI as a tool to study the role of kappa opioid receptors relating to animal behavior and psychosis models.
In conclusion, our current study using GNTI showed that it can decrease MK-801-induced stereotypy and hyperlocomotion. The pharmacological profile of GNTI as a selective kappa opioid antagonist thus links the kappa opioid receptor in the modulation of MK-801-induced behavior, pointing to a potential route of developing novel antipsychotic drugs to treat schizophrenia and other mental illnesses.
References
- Fleur LS. Neuropeptides: regulators of physiological process. Cambridge, MA: The MIT Press; 1999.
- Zou G. Basic nerve pharmacology. Beijing: Science Press; 1999.
- Smith JA, Loughlin SE, Leslie FM. kappa-Opioid inhibition of [3H]dopamine release from rat ventral mesencephalic dissociated cell cultures. Mol Pharmacol 1992;42:575-83.
- Chefer VI, Shippenberg TS. Paradoxical effects of prodynorphin gene deletion on basal and cocaine-evoked dopaminergic neurotransmission in the nucleus accumbens. Eur J Neurosci 2006;23:229-38.
- Marin C, Bove J, Serrats J, Cortes R, Mengod G, Tolosa E. The kappa opioid agonist U50,488 potentiates 6-hydroxydopamine-induced neurotoxicity on dopaminergic neurons. Exp Neurol 2005;191:41-52.
- Margolis EB, Hjelmstad GO, Bonci A, Fields HI. kappa-Opioid agonists directly inhibit midbrain dopaminergic neurons. J Neurosci 2003;23:9981-6.
- Jones RM, Hjorth SA, Schwartz TW, Portoghese PS. Mutational evidence for a common kappa antagonist binding pocket in the wild-type kappa and mutant mu[K303E] opioid receptors. J Med Chem 1998;41:4911-4.
- Tortella FC, Robles L, Echevarria E, Hunter JC, Hughes J. PD117302, a selective non-peptide opioid kappa agonist, protects against NMDA and maximal electroshock convulsions in rats. Life Sci 1990;46:PL1-7.
- Singh L, Vass CA, Hunter JC, Woodruff GN, Hughes J. The anticonvulsant action of CI-977, a selective kappa-opioid receptor agonist: a possible involvement of the glycine/NMDA receptor complex. Eur J Pharmacol 1990;191:477-80.
- De Sarro G, Trimarchi GR, Sinopoli S, Masuda Y, De Sarro A. Anticonvulsant effects of U-54494A and U-50488H in genetically epilepsy-prone rats and DBA/2 mice: a possible involvement of glycine/NMDA receptor complex. Gen Pharmacol 1993;24:439-47.
- Izenwasser S, Staley JK, Cohn S, Mash DC. Characterization of kappa1-opioid receptor binding in human insular cortex. Life Sci 1999;65:857-62.
- Royston MC, Slater P, Simpson MD, Deakin JF. Analysis of laminar distribution of kappa opiate receptor in human cortex: comparison between schizophrenia and normal. J Neurosci Methods 1991;36:145-53.
- Pogozheva ID, Przydzial MJ, Mosberg HI. Homology modeling of opioid receptor-ligand complexes using experimental con-straints. AAPS J 2005;7:E434-48.
- Metcalf MD, Coop A. Kappa opioid antagonists: past successes and future prospects. AAPS J 2005;7:E704-22.
- Hiramatsu M, Watanabe M, Baba S, Kojima R, Nabeshima T. Alpha 7-type nicotinic acetylcholine receptor and prodynorphin mRNA expression after administration of (-)-nicotine and U-50,488H in beta-amyloid peptide (25-35)-treated mice. Ann N Y Acad Sci 2004;1025:508-14.
- Jones RM, Portoghese PS. 5'-Guanidinonaltrindole, a highly selective and potent kappa-opioid receptor antagonist. Eur J Pharmacol 2000;396:49-52.
- Negus SS, Mello NK, Linsenmayer DC, Jones RM, Portoghese PS. Kappa opioid antagonist effects of the novel kappa antagonist 5'-guanidinonaltrindole (GNTI) in an assay of schedule-controlled behavior in rhesus monkeys. Psychopharmacology (Berl) 2002;163:412-9.
- Cook CD, Newman JL, Winfree JC, Beardsley PM. Modulation of the locomotor activating effects of the noncompetitive NMDA receptor antagonist MK801 by dopamine D2/3 receptor agonists in mice. Pharmacol Biochem Behav 2004;77:309-18.
- Wu J, Zou H, Strong JA, Yu J, Zhou X, Xie Q, et al. Bimodal effects of MK-801 on locomotion and stereotypy in C57BL/6 mice. Psychopharmacology 2005;177:256-63.
- Wu JH, Zou H, Yu J, Zhou XD, Xie QL, Jin ML. Animal models of schizophrenia using different laboratory mouse strains. Acta Physiol Sin 2003;55:381-7.
- Mozayani A, Schrode P, Carter J, Danielson TJ. A multiple drug fatality involving MK-801 (dizocilpine), a mimic of phencycli-dine. Forensic Sci Int 2003;133:113-7.
- Jewett DC, Grace MK, Jones RM, Billington CJ, Portoghese PS, Levine AS. The kappa-opioid antagonist GNTI reduces U50,488-, DAMGO-, and deprivation-induced feeding, but not butorphanol- and neuropeptide Y-induced feeding in rats. Brain Res 2001;909:75-80.
- Feng J, Kendig JJ. The NMDA receptor antagonist MK-801 differentially modulates mu and kappa opioid actions in spinal cord in vitro. Pain 1996;66:343-9.
- Peckys D, Hurd YL. Prodynorphin and kappa opioid receptor mRNA expression in the cingulate and prefrontal cortices of subjects diagnosed with schizophrenia or affective disorders. Brain Res Bull 2001;55:619-24.
- Horan P, Taylor J, Yamamura HI, Porreca F. Extremely long-lasting antagonistic actions of nor-binaltorphimine (nor-BNI) in the mouse tail-flick test. J Pharmacol Exp Ther 1992;260:1237-43.
- Hlinak Z, Krejci I. Spontaneous alternation behaviour in rats: kynurenic acid attenuated deficits induced by MK-801. Behav Brain Res 2006;168:144-9.
- Tang XZ, Cui NJ. Clinical biochemistry. Tianjin: Tianji Science and Technology Press; 1999.
- Portoghese PS, Lipkowski AW, Takemori AE. Binaltorphimine and nor-binaltorphimine, potent and selective kappa-opioid receptor antagonists. Life Sci 1987;40:1287-92.
- McLaughlin JP, Marton-Popovici M, Chavkin C. Kappa opioid receptor antagonism and prodynorphin gene disruption block stress-induced behavioral responses. J Neurosci 2003;23:5674-83.
- Su MT, Lin WB, Lue WM, Cheng CY, Tao PL. Blockade of the development of morphine tolerance by U-50,488, an AVP antagonist or MK-801 in the rat hippocampal slice. Br J Pharmacol 1998;123:625-30.
- Bortolato M, Aru GN, Frau R, Orru M, Fa M, Manunta M, et al. Kappa opioid receptor activation disrupts prepulse inhibition of the acoustic startle in rats. Biol Psychiatry 2005;57:1550-8.
- Sinchaisuk S, Ho IK, Rockhold RW. Focal kappa-opioid receptor-mediated dependence and withdrawal in the nucleus paragigantocellularis. Pharmacol Biochem Behav 2002;74:241-52.
- Savitsky A, Golay MJE. Smoothing and differentiation of data by simplified lest square procedures. Analytical Chemistry 1964;36:1627-39.
